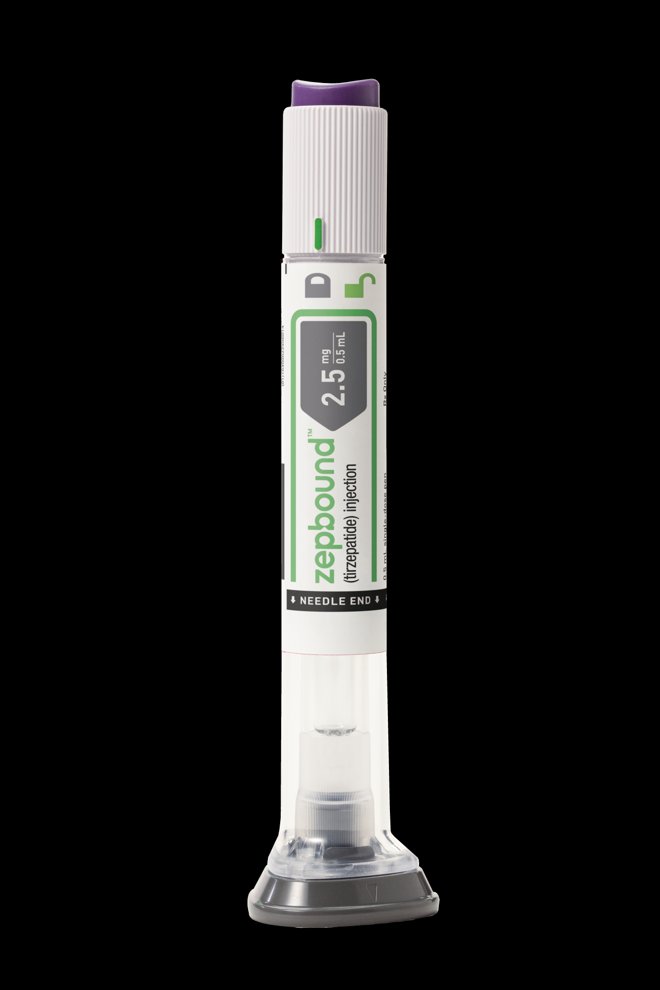
Tirzepatide approved for obesity by the FDA – to be branded as Zepbound
Eli Lilly’s drug with the active ingredient tirzepatide has already been approved for treating type 2 diabetes. The drug has gained publicity because it causes significant weight loss, and now the US Food and Drug Administration (FDA) has approved the same substance for obesity.
According to the FDA, the approval is for patients who are obese or overweight and have at least one condition related to obesity. When taken for obesity, the drug will be marketed under the brand name Zepbound.
Tirzepatide works by activating receptors for the hormones GIP and GLP-1. The latter is also the target of Novo Nordisk’s well-known diabetes and obesity drug Ozempic/Wegovy (semaglutide).
Zepbound is expected to be available in the US before the end of the year and at a price lower than semaglutide, Eli Lilly states in a press release.
Artikeln är en del av vårt tema om News in English.
 Av
Av